Homemade production of bleach disinfectant
… otherwise known chlorine, bleach is today one of the best know disinfectants and is commonly used to treat drinking water in european water main.
The proposed method makes use of basic materials that can be found easily, anywhere.
Properties
Kills off effectively, in just a few minutes, the bacillus of:
tetanus, cholera, typhus, carbuncle, hepatitis, entero virus,
streptococcus, staphylococcus, etc.
Uses:
- to disinfect drinking water
- to disinfect vegetables and kitchen ware (plates, pots and pans, cutlery)
- to sterilize feeding bottle
- to sterilize clothes, sheets, walls, floors (homes and hospitals)
- to disinfect cuts, sores, etc.
- to disinfect surgical instruments
- to disinfect animals, stable walls, etc
- to disinfect excrements
How to produce bleach at home
Materials:
- a tall plastic, 1.5 bottle with screw top
- a cylindrical charcoal stick taken from a large electrical torch (N.B.
batteries with a plastic ring contain a carbon electrode while other batteries have a
metal ring, and so being alkaline, they have a metal electrode) - a wax candle
- a container
- kitchen salt or for animal feed
- water (preferably rain water or filtered water)
- a container in which to mix salt and water
- an electric current of 12 to 24 Volts (such as a car, jeep, lorry battery
or even photovoltaic sun panels)
Procedure
Step one: preparing the materials
- cut off the bottom of the plastic bottle
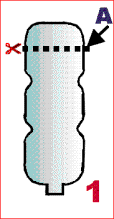
A- where cut off
- take the charcoal out the torch with tweezers. With a candle flame, melt away any wax residues. Clean the charcoal with a piece of paper.
- make a hole in the bottle’s screw top, big enough in order for the
charcoal stick to be inserted; if necessary, seal the charcoal stick with a few drops of candle wax;
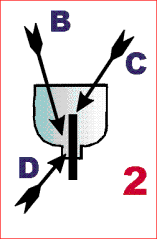
B the candle wax
C the charcoal,
D the bottle top
- screw the bottle top back on;
- Take 1.5 litres of water and add a handful of salt (about 100 gr.)Mix well until the salt is entirely dissolved into the water.Pour the solution into the prepared bottle – right up to the top
|
picture 4 Connect the tip and the negative (black) electrode, without the plastic |
 |
What happens
 |
From the charcoal bubbles of gas will develop (chlorine)thet will then transform the salty water into disinfectant bleach. The smaller the bubbles, the the better reaction. The wider the diameter of the carbon stick, the smaller the bubbles will be.Even the black wire will produce bubbles but they will be of hydrogen and will bot be of any use..After about one hour of reaction, the bleach will be concentrate enough. The reaction can be left for up to two hours. By then the liquid solution will be yellowish and will produce the typical smell of chlorine..picture 5 G battery H chlorine bubbles I hydrogen bubbles |
General formula:
| 1 handful of salt + 1 litre of water + 1 hour = 1 litre bleach |
Here is an example of how to make water drinkable:
To disinfect 5 litres of water add enough bleach to get a slight taste
of chlorine (about 2 to 3 tablespoons) mix very, very well – because it’s the contact of
the chlorine with the water molecules that disinfect, and not the density of chlorine in
the water.
Try tasting the water again after about 20 minutes. You may add some
more bleach to get the slight chlorine taste – this will ensure the water remains sterile
until its use. Remember, chlorine will be consumed in the presence of bacteria and
micro-organisms.
N. :
Una risposta